- Blog
- Setup fishbowl inventory
- Modbus poll piratebay
- Rurouni kenshin kyoto inferno english sub watch online
- Pokemon gba rom hack pokemon unbonud
- N lab master
- Trimble survey controller manual
- Bentley water cad free trial
- Pokkiri raja tamil movie jeeva
- Virtual stripper game
- Nero vision express mac
- Nonpolar hydrophobic amino acids
- Algebra de mancil tomo 1 ejercicios 86 resueltos
- Un dolor imperial descargar
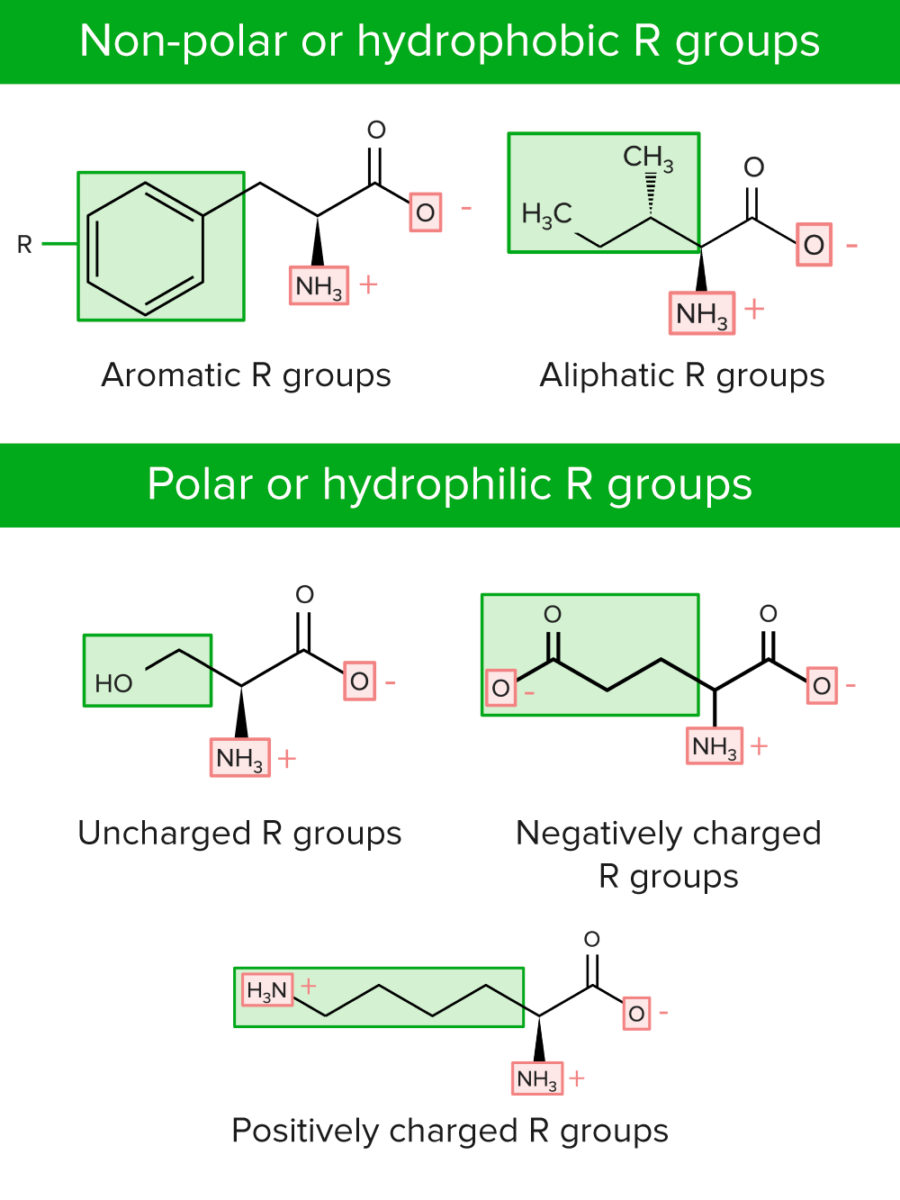
1 Near a solid surface, proteins tend to adsorb, and depending on the protein and surface properties, they may undergo partial or significant conformational changes. Introduction When a protein is placed outside its natural medium, its structure and function are usually affected. Electronic calculations carried out on rebuilt all-atom coordinates of adsorbed lysozymes revealed consistent destabilization of helices, while beta sheets show a greater variety of trends. Differences between helical and sheet structures are observed upon denaturation – α-helices undergo spreading of their native helical order to an elliptical perturbed shape, while β-sheets transform into random coils and other more structured conformations. The hydrophobic scale BULDG was chosen for further analysis due to its greater stability during heating and its partial regenerative ability upon slow cooling. Three different hydrophobicity scales are tested for adsorption of fibronectin on a hydrophobic surface. Intermolecular interactions are modeled using the Miyazawa–Jernigan (MJ) knowledge-based potential for an implicit solvent. Hydrophobicity scales can also be obtained by calculating the solvent accessible surface areas for amino acid residues in the expended polypeptide chain or in alpha-helix and multiplying the surface areas by the empirical solvation parameters for the corresponding types of atoms.This paper presents a study of protein adsorption and denaturation using coarse-grained Monte Carlo simulations with simulated annealing. How do you know if amino acid is hydrophobic? Tyrosine is the only one of the aromatic amino acids with an ionizable side chain. Tyrosine and tryptophan absorb more than do phenylalanine tryptophan is responsible for most of the absorbance of ultraviolet light (ca. You can tell if a protein is hydrophobic or hydrophilic by examining the side chains of amino acids in its sequence. How do you know if a protein is hydrophobic or hydrophilic? 5-6), are relatively nonpolar (hydrophobic). Aromatic R Groups Phenylalanine, tyrosine, and tryptophan, with their aromatic side chains (Fig. The secondary amino (imino) group is held in a rigid conformation that reduces the structural flexibility of the protein at that point.

It helps in the function of organs responsible for making and regulating hormones, including the adrenal, thyroid, and pituitary glands.
#Nonpolar hydrophobic amino acids skin
Tyrosine also helps produce melanin, the pigment responsible for hair and skin color. Neurotransmitters help nerve cells communicate and influence mood. For example, even phenylalanine only has an aromatic ring, tyrosine is more soluble than it in water. The hydroxyl group attached to the aromatic gives it a polar characteristic while its aromatic ring gives it the hydrophobic characteristic. You might be interested: Question: How long can a moth live? Why is phenylalanine more hydrophobic than tyrosine? 2.0 mg/ml at pH 9.5, the solubility is 1.4 mg/ml and at pH 10, the solubility is 3.8 mg/ml. The solubility in water (25 ☌) is 0.45 mg/ml in the pH range 3.2 – 7.5. This product is soluble in 1 M HCl (100 mg/ml), with heating. IMGT classes of the amino acids side chain properties Is phenylalanine hydrophilic or hydrophobic? The hydroxyl groups in these three amino acids are subject to an important type of posttranslational modification: phosphorylation (see below Nonstandard amino acids). Group II: Polar, uncharged amino acids Tyrosine possesses a hydroxyl group in the aromatic ring, making it a phenol derivative. There are only five atoms that will appear in your amino acid variable groups: H, C, N, O, and S. The lack of polarity means they have no way to interact with highly polar water molecules, making them water fearing. Hydrophobic amino acids have little or no polarity in their side chains.
- Blog
- Setup fishbowl inventory
- Modbus poll piratebay
- Rurouni kenshin kyoto inferno english sub watch online
- Pokemon gba rom hack pokemon unbonud
- N lab master
- Trimble survey controller manual
- Bentley water cad free trial
- Pokkiri raja tamil movie jeeva
- Virtual stripper game
- Nero vision express mac
- Nonpolar hydrophobic amino acids
- Algebra de mancil tomo 1 ejercicios 86 resueltos
- Un dolor imperial descargar
